Vijayagopal Gopalsamuthiram*, Appasaheb L. Kadam, Jeffrey K. Noble, David R. Snead, Corshai Williams, Timothy F. Jamison, Chris Senanayake (TCG Green Chem Member), Ajay K. Yadaw (TCG Green Chem Member), Sarabindu Roy (TCG Green Chem Member), Gopal Sirasani (TCG Green Chem Member), B. Frank Gupton, Justina Burns, Daniel W. Cook, Rodger W. Stringham, Saeed Ahmad, and Rudy Krack
Organic Process Research and Development (Published on 09th Dec 2021)
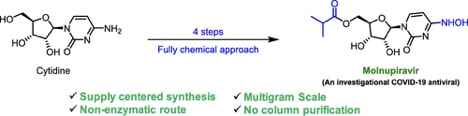
Abstract: A scalable four-step synthesis of molnupiravir from cytidine is described herein. The attractiveness of this approach is its fully chemical nature involving inexpensive reagents and more environmentally friendly solvents such as water, isopropanol, acetonitrile, and acetone. Isolation and purification procedures are improved in comparison to our earlier study as all intermediates can be isolated via recrystallization. The key steps in the synthesis, namely, ester formation, hydroxyamination, and deprotection were carried out on a multigram scale to afford molnupiravir in 36–41% yield with an average purity of 98 wt % by qNMR and 99 area% by HPLC.